OUR NEW PROFILE IS: (NASDAQ: SILO)
- Recently SILO announced its novel liposomes reached a positive endpoint in animal studies showing effectiveness and reduced toxicity using a subcutaneous delivery method
- Recently SILO announced its topically administered Ketamine reached a positive end point in an animal study.
- SILO uplisted to Nasdaq on September 27 and completed a $5.7 million public offering
- SILO has $8.7 million in the treasury and is virtually debt free according to their 10Q filed on August 1
- SILO entered into A sponsored study with Maastricht University utilizing repeated low doses of ketamine and psilocybin to examine the effects on cognitive and emotional dysfunctions in Parkinson’s disease;
- SILO entered into A scientific research agreement with the University of California San Francisco (UCSF) leveraging four other clinical trials being planned by the university to determine the effects of psilocybin on inflammation;
- SILO signed an exclusive license agreement with the University of Maryland, Baltimore (UMB) to explore a patent focused on enhanced targeting of therapeutic agents to the central nervous system;
- SILO signed a second exclusive option agreement with UMB to license a joint-homing peptide targeted at arthritis-inflamed joints
- SILO entered into an agreement with Columbia University granting it an option to license two distinct assets currently under development, one focused on Alzheimer’s disease and a second focused on PTSD and stress.
Check out the Company website here: silopharma.com
SILO is moving steadily northbound since reversing off established $4.50 support level.
_______________________
Hello Everyone,
The market is set to open and we have something that we feel could be super explosive immediately.
This on is sitting at $5.38 right now down from over $12 earlier this month.
Turn your attention to SILO right away.
Silo Pharma Inc (NASDAQ: SILO) is a developmental stage biopharmaceutical company focused on merging traditional therapeutics with psychedelic research. Silo is committed to developing innovative solutions to address conditions such as post-traumatic stress disorder (PTSD), fibromyalgia, Alzheimer’s disease, Parkinson’s disease, and other rare neurological disorders. The company works to identify and partner with leading medical universities, providing the needed financial resources to develop safe therapeutic treatments while moving cutting-edge research through the clinical stage and into commercialization.
Intellectual property: To date, Silo has filed 4 provisional patent applications related to the use of the central nervous system-homing peptides covered by the UMB Option Agreement to deliver certain compounds, including a nonsteroidal anti-inflammatory drug and/or psilocybin, for the treatment of arthritis, central nervous system diseases, neuroinflammatory diseases as well as cancer. In addition, pursuant to the Company’s acquisition of NFID, Silo acquired three trademarks related to the NFID brand.
SILO is Harnessing the Power of Psychedelics to Deliver Breakthrough Medical Solutions
Silo has entered into research agreements and partnerships with multiple leading medical universities, including:
- A sponsored study with Maastricht University utilizing repeated low doses of ketamine and psilocybin to examine the effects on cognitive and emotional dysfunctions in Parkinson’s disease;
- A scientific research agreement with the University of California San Francisco (UCSF) leveraging four other clinical trials being planned by the university to determine the effects of psilocybin on inflammation;
- An exclusive license agreement with the University of Maryland, Baltimore (UMB) to explore a patent focused on enhanced targeting of therapeutic agents to the central nervous system;
- A second exclusive option agreement with UMB to license a joint-homing peptide targeted at arthritis-inflamed joints; and
- An agreement with Columbia University granting it an option to license two distinct assets currently under development, one focused on Alzheimer’s disease and a second focused on PTSD and stress.
Psilocybin is considered a serotonergic hallucinogen and is an active ingredient in some species of mushrooms. Recent industry studies using psychedelics, such as psilocybin, have been promising, and Silo Pharma believes there is a large unmet need with many people suffering from depression, mental health issues and neurological disorders. While classified as a Schedule I substance under the Controlled Substances Act (“CSA”), there is an accumulating body of evidence that psilocybin may have beneficial effects on depression and other mental health conditions. Therefore, the U.S. Food and Drug Administration (“FDA”) and U.S. Drug Enforcement Agency (“DEA”) have permitted the use of psilocybin in clinical studies for the treatment of a range of psychiatric conditions.
The potential of psilocybin therapy in mental health conditions has been demonstrated in a number of academic-sponsored studies over the last decade. In these early studies, it was observed that psilocybin therapy provided rapid reductions in depression symptoms after a single high dose, with antidepressant effects lasting for up to at least six months for a number of patients. These studies assessed symptoms related to depression and anxiety through a number of widely used and validated scales. The data generated by these studies suggest that psilocybin is generally well-tolerated and has the potential to treat depression when administered with psychological support.
Silo Pharma has engaged in discussions with a number of world-renowned educational institutions and advisors regarding potential opportunities and have formed a scientific advisory board that is intended to help advise management regarding potential acquisition and development of products.
In addition, as more fully described below, Silo Pharma has entered into a license agreement with the University of Baltimore, Maryland, and has entered into a joint venture with Zylo Therapeutics, Inc., with respect to certain intellectual property and technology that may be used for targeted delivery of potential novel treatments. In addition, Silo Pharma has recently entered into a sponsored research agreement with Columbia University pursuant to which it has been granted an option to license certain patents and inventions relating to the treatment of Alzheimer’s disease and stress-induced affective disorders using Ketamine in combination with certain other compounds.
Silo Pharma plans to actively pursue the acquisition and/or development of intellectual property or technology rights to treat rare diseases, and to ultimately expand our business to focus on this new line of business.
Silo Pharma Announces Positive Results from its Topically Administered Formulation of Ketamine
Silo shows reversal of allodynia and hyperalgesia using its selected formulation and partnered, patented delivery system
ENGLEWOOD CLIFFS, N.J, July 27, 2022 (GLOBE NEWSWIRE) — Silo Pharma, Inc . (OTCQB: SILO), a development-stage biopharmaceutical company today announced that its topically administered Ketamine reached a positive end point in an animal study.
Silo’s formulation reduced mechanical allodynia and hyperalgesia at both the pre-and post-dosing time points, and mechanical hyperalgesia was reduced on day seven at the pre-dose time point. These results indicate that the dosing of Ketamine using a patent protected delivery system with Silo Pharma’s partner, Zylo Therapeutics, yielded positive results in reducing neuropathic nerve pain in a small animal study.
Eric Weisblum, CEO of Silo Pharma, commented, “These positive results of our study show that Silo is able to successfully formulate and deliver ketamine topically at reduced dosage to achieve its endpoint of pain reduction. This data is extremely promising for patients suffering from Fibromyalgia and Rheumatoid Arthritis. Additionally, I am delighted to report that Day seven and Day nine scores averaged zero (no erythema or swelling present) for all groups, which bodes extremely well for our planned toxicology study.
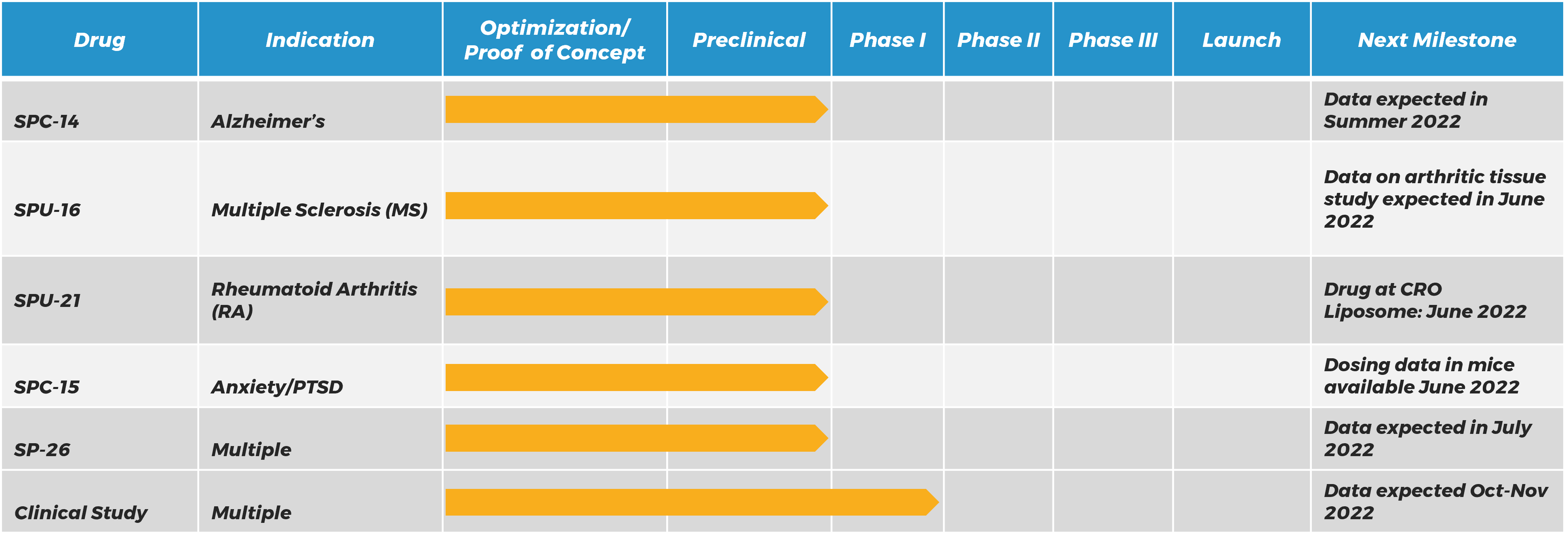
SILO Pipeline:
Silo Pharma believes in following the science to identify the best application for a therapeutic rather by targeting indications with the biggest need and selecting the best route of delivery for patients. Our current assets have shown promise in a range of illnesses and diseases, including Alzheimer’s; Parkinson’s; Multiple Sclerosis (MS); Rheumatoid Arthritis (RA); and Stress-Induced Psychiatric Disorders.
SILO Pharma’s Primary Asset Has Extraordinary Potential
505(b)(2) Pathway / preclinical testing and proof-of-concept being lead by inventor Dr. Christine Denny of Columbia University / SPC-14 has shown reduced hyponeophagia in animal studies / SPC-14 may reduce behavioral despair / Silo has licensed technology with Columbia and has recently entered into a scientific research agreement with Dr. Denny’s lab
Market Opportunity – Alzheimer’s Disease / 6.5 million Americans suffer from Alzheimer’s and related diseases / ~1 in 9 Americans 65+ have Alzheimer’s v U.S. market for relevant drugs expected to reach $5 billion by 2027
Technology – Licensed from Columbia University / SPC-14 is a novel drug combining two approved therapeutics / SPC-14 targets NDMARS and 5HT4Rs to treat cognitive and neuropsychiatric symptoms in Alzheimers.
 SPC-15 Targeted prophylactic treatment—Stress-induced affective disorders
SPC-15 Targeted prophylactic treatment—Stress-induced affective disorders
Sponsored Research Agreement with Columbia University Prevention of stress-induced affective disorders / Increasing stress resilience in military, first responders, and other populations at high risk of PTSD / Predicting the level of severity or progression such disorders / Molecular targets for use in drug discovery of innovative treatments.
Market Opportunity – Targeted prophylactic treatment—Stress-induced affective disorders / 26% of Americans 18+ suffer from anxiety, PTSD and other disorders / This number has escalated post-COVID-19 / U.S. market for relevant drugs expected to reach $13 billion by 2027
Technology – Metabolomic biomarkers predict response to pharmacological treatments / Utilizes ketamine compositions as a method for treatment and prevention.
 SP-26 Time-Released Psilocybin, Ketamine
SP-26 Time-Released Psilocybin, Ketamine
Deliver Ketamine or Psilocybin in a time-released manner / Will time-release diminish the hallucinogenic effects of these psychedelics / Pre clinical study underway shows Z-pod can hold and distribute Ketamine / Efficacy study in animals underway.
Market Opportunity – Time-Released Psilocybin, Ketamine / Multiple Indications
Technology – Joint Venture with Zylö Therapeutics, Inc. / Clinical development of psilocybin using ZTI’s Z-pod technology / Clinical development of Zylo’s sustained release topical delivery system.
May be used as a delivery tool to target current therapies to detect inflammation in the spinal cord / May be used for diagnosing and monitoring MS / Decreases toxicity of existing therapeutics / Animal study results show much improved delivery of therapeutics and decreased toxicity.
Market Opportunity – Multiple Sclerosis (MS) / There are approximately 400,000 Americans and 2.5 million people worldwide with MS / The most widespread disabling neurological condition of young adults / Global market for MS drugs expected to reach $25.3 billion by 202
Technology- Licensed from University of Maryland Baltimore / Patent issued / Central nervous system-homing peptides / Use for investigation and treatment of MS and other neuroinflammatory pathology.
 SPU-21 Arthritogenic Joint Homing Peptides Utilizing Psilocybin
SPU-21 Arthritogenic Joint Homing Peptides Utilizing Psilocybin
Identify markers of arthritic inflammation in joints / Isolate phage clones that preferentially target inflamed joints of arthritic Lewis rats / Peptide significantly inhibited arthritic progression in this animal model / Further studies are underway at UMB.
Market Opportunity – Rheumatoid Arthritis (RA) / 1.3M U.S. adults suffer from RA / The most common autoimmune disease in U.S. / U.S. market for RA drugs expected to reach $63 billion by 2027
Technology- Development plan to utilizing liposomal Homing Peptide to deliver targeted psilocybin / The ability of the peptides to target inflamed epithelium suggest they could be used to target drug delivery. This approach could enhance the therapeutic effect of current and future therapies and decrease potential systemic toxicity despite systemic administration of the drug. These peptides have potential for the development of fusion imaging molecules and/or nanoparticles to study arthritic pathogenesis. They could also be customizable and used to deliver nanoparticles for precise imaging. In addition, these novel joint-homing peptides can be used to treat autoimmune diseases, including but not limited to RA.
In July SILO announced its novel liposomes reached a positive endpoint in animal studies showing effectiveness and reduced toxicity using a subcutaneous delivery method. In previous work, ART-1-Cy7-liposomes injected i.v. demonstrated homing to arthritic joints and 4 hours was an optimal time point for high fluorescence intensity in the hind paws. The results of this new study show that the time kinetics and quantitative aspects of liposomes administered subcutaneous were not much different from that of liposomes injected i.v. In fact, despite some initial delay, the signal strength showed a trend towards a higher level in subcutaneous than in i.v.
Eric Weisblum, CEO of Silo Pharma commented “The successful results of our study show that the subcutaneous route of liposomal administration is well-suited for use in targeted drug delivery of anti-arthritic agents. We suggest that this drug delivery approach would not only enhance efficacy but also reduce systemic toxicity.”
Also, in July SILO announced its topically administered Ketamine reached a positive end point in an animal study. Silo’s formulation reduced mechanical allodynia and hyperalgesia at both the pre-and post-dosing time points, and mechanical hyperalgesia was reduced on day seven at the pre-dose time point. These results indicate that the dosing of Ketamine using a patent protected delivery system with Silo Pharma’s partner, Zylo Therapeutics, yielded positive results in reducing neuropathic nerve pain in a small animal study.
Eric Weisblum, CEO of Silo Pharma, commented, “These positive results of our study show that Silo is able to successfully formulate and deliver ketamine topically at reduced dosage to achieve its endpoint of pain reduction. This data is extremely promising for patients suffering from Fibromyalgia and Rheumatoid Arthritis. Additionally, I am delighted to report that Day seven and Day nine scores averaged zero (no erythema or swelling present) for all groups, which bodes extremely well for our planned toxicology study.”
How SILO Pharma’s Drug Delivery Platform Could Cause Massive Changes to How Diseases Are Treated
Research has clearly shown that psychedelics – such as psilocybin – can offer significant benefits to those suffering from a number of disorders. But to date, the issue has been developing a psychedelic-based solution for these sufferers that was both practical and effective. Thanks to the potential of SILO Pharma’s unique drug delivery platform – developed in conjunction with university research – we could be on the verge of a significant breakthrough in treatment. SILO Pharma’s assets and research could be transformative to the well-being of patients worldwide…and disruptive to the healthcare industry as a whole. That alone sets Silo Pharma (OTC: SILO) apart from others in both the psychedelic and biopharma arenas.
The research and development – based on the company’s unique homing peptide – could not only prove to be effective in the delivery of psychedelics…but also in delivery of traditional medication. That type of potential is deserving of attention from investors…but it’s also more difficult to project. A potential comparable stock that has demonstrated the potential for this company’s upside, however, does exist in the cannabis space.
1) Double Play Investment Opportunity – Unlike many companies generating attention in the psychedelic space, SILO Pharma is focused on developing potential breakthroughs by combining traditional therapeutics with the power of psychedelic research.
2) Rapidly Growing Market – The market for psychedelics is projected to grow at a CAGR of 16.3% between now and 2027, when it is expected to reach $6.8 billion.
3) Potential Game-Changing Drug Delivery Platform – SILO Pharma’s potentially game-changing drug delivery platform may be used to treat a number of devastating diseases, including Rheumatoid Arthritis and Multiple Sclerosis and is likely to generate considerable attention from larger companies, making for attractive buyout potential.
4) Following a Proven Blueprint for Success – The company is following the same path forged in the cannabis sector by GW Pharma, and is working to combine the powerful healing benefits of psilocybin with its own unique drug delivery system in the hopes of developing a true game-changer that could ultimately prove disruptive to the entire healthcare industry.
5) Proven Leadership Team – Silo Pharma is led by an experienced team of business builders and researchers poised to make the company’s dream a reality. This team includes CEO Eric Weisblum, with over 20 years investing, building and managing businesses as well as an impressive scientific advisory board.
SILO has a compelling chart setup: SILO has been upward bound since reversing northbound off established $4.50 support levels. The last time it tested this support levels it more than doubled in the preceding months.
The Psychedelics Market is Experiencing Rapid Growth:
 Thanks to great research showing the healing power of psychedelic drugs – such as psilocybin – the psychedelics market is experiencing rapid growth.
Thanks to great research showing the healing power of psychedelic drugs – such as psilocybin – the psychedelics market is experiencing rapid growth.
In fact, experts project that this market will grow at an impressive compound annual growth rate (CAGR) of 16.3% between now and 2027.[i]
This projected growth will mean the market will more than triple from its current size of $2.077 billion to more than $6.8 billion by 2027.
As new research continues to emerge about the efficacy of psilocybin and other psychedelics, investors are seeing a number of companies emerge in an attempt to establish an early leadership position in the space. But as these companies jockey for position, it’s important to look closely at what each “mushroom stock” brings to the table for potential investors. Many up-and-coming companies may have strong financial backing – or aggressive business plans – but lack one critical element: intellectual property.
That’s where one company – currently flying under the radar of most investors – appears to have a distinct advantage.
SILO Recent News:
 Silo Pharma Announces Uplisting to Nasdaq Capital Market and Pricing of $5 Million Public Offering of Common Stock
Silo Pharma Announces Uplisting to Nasdaq Capital Market and Pricing of $5 Million Public Offering of Common Stock
Press Release | 09/27/2022
ENGLEWOOD CLIFFS, NJ , Sept. 27, 2022 (GLOBE NEWSWIRE) — Silo Pharma, Inc. (Nasdaq: SILO) (formerly OTCQB: SILO), a developmental stage biopharmaceutical company focused on merging traditional therapeutics with psychedelic research, today announced pricing of an underwritten public offering of 1,000,000 shares of its common stock at a price to the public of $5.00 per share.
The gross proceeds to Silo Pharma from this offering are expected to be $5,000,000, before deducting the underwriting discount and other estimated offering expenses payable by Silo Pharma. Silo Pharma intends to use the net proceeds from the offering for product development, marketing, and working capital and general corporate purposes. A portion of the proceeds may also be used for acquisitions of complementary businesses, technologies, or other assets.
As a result of the offering, the Company’s common stock will become listed on the Nasdaq Capital Market and will trade under the ticker symbol “SILO” beginning September 27, 2022. The offering is expected to close on or about September 29, 2022, subject to customary closing conditions. In addition, the Company has granted a 45-day option to the underwriter to purchase up to an additional 150,000 shares of common stock at the public offering price, less the underwriting discount, to cover over-allotments, if any.
Laidlaw & Company (UK) Ltd. is acting as sole book-running manager for the offering.
A registration statement on Form S-1 (File No. 333-261532) relating to the public offering of the shares of common stock described above was previously filed with the Securities and Exchange Commission (“SEC”) and declared effective on September 15, 2022. The final prospectus will be filed with the SEC and will be available on the SEC’s website at www.sec.gov. Copies of the final prospectus, when available, may be obtained from Laidlaw & Company (UK) Ltd., 521 Fifth Ave., 12th Floor, New York, NY 10175, Attention: Syndicate Dept.; email: syndicate@laidlawltd.com.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities laws of any such state or other jurisdiction. This offering is being made only by means of the prospectus.
SILO RECENT NEWS
SILO EXECUTIVE TEAM
Silo Pharma’s is Led by an Experienced Management Team and an Impressive Group of Scientific Advisors
 CEO Eric Weisblum has over 20 years investing, building and managing businesses and will lead a scientific advisory board of reputable medical professionals to work and guide the company through the different stages of research, licensing, partnerships and trials.
CEO Eric Weisblum has over 20 years investing, building and managing businesses and will lead a scientific advisory board of reputable medical professionals to work and guide the company through the different stages of research, licensing, partnerships and trials.
Prior, Mr. Weisblum was President of Sableridge Capital for five years. Eric currently serves on the board of directors of Aikido Pharma., a Nasdaq listed biotech company focused on the commercialization of oncology therapeutics. In addition to being an active investor in both public and private companies, Mr. Weisblum has provided managerial assistance and guidance to help companies execute on their business strategy.
 Director Wayne Linsley has been an entrepreneur for over 40 years. In 1979, he received a Bachelor’s Degree in Business Administration from Sienna College in Loudonville, New York.
Director Wayne Linsley has been an entrepreneur for over 40 years. In 1979, he received a Bachelor’s Degree in Business Administration from Sienna College in Loudonville, New York.
He has since been involved with real estate brokerage and residential development, construction, finance, telecommunications. Since 2009 he has worked for and is currently a Vice President for CFO Oncall, Inc., a financial reporting firm that works with publicly traded companies.
 Director Kevin Munoz, MD, currently serves as Director of Operations at Physical Medicine and Rehabilitation where he has responsibility for the day-to-day management of all office operations with a focus on ensuring and increasing patient satisfaction. Prior to that, he led the configuration efforts during an enterprise-wide implementation of application software that also included streamlining and improving business processes.
Director Kevin Munoz, MD, currently serves as Director of Operations at Physical Medicine and Rehabilitation where he has responsibility for the day-to-day management of all office operations with a focus on ensuring and increasing patient satisfaction. Prior to that, he led the configuration efforts during an enterprise-wide implementation of application software that also included streamlining and improving business processes.
Sincerely,

DISCLAIMER
Sincerely,
The Viral Stocks Team